AFM and SEM Imaging of Bone
A major part of our research into the molecular origins of fracture mechanisms in bone is imaging bone with the Atomic Force Microscope (AFM) and the Scanning Electron Microscope (SEM). The ever-increasing, extremely high viewing resolution that can be achieved with the AFM and the SEM open up an increasingly deep view of how living (and other) structures work.
The following images, which include some of the highest-resolution images ever taken of bone, have been taken by our group members, often in interdisciplinary collaboration with other labs at UCSB, such as the Dan Morse, Herb Waite and Galen Stucky labs. All images that appear on this page have been published in our papers or are in press.
Bone ultrastructure and bone glue
Before bone ultrastructure could be viewed non-destructively with the AFM, the ultrastructure of bone and bone building blocks - collagen fibrils, mineral plates and bone's non-fibrillar protein-based organic matrix - could not be seen as accurately as with the AFM. This is because other forms of non-optical microscopy require preparing and imaging delicate biological samples in destructive artificial conditions, such as metal plating, extreme cold or high vacuum. Therefore, characteristics of bone building blocks, such as the size and shapes of mineral particles of bone, were also estimated through theoretical computations. The AFM can reveal biological structures and processes as they are - depending, of course, on what is studied (imaging very fast biological processes on the order of micro- and nanoseconds is not presently in the AFM's reach, and may never be) and depending on the corresponding capabilities of the AFM used. We do believe that current AFMs operate far beneath their fundamental limits, so fast enough biological processes and small enough biological features are too small and fast for current AFMs to capture. The development of ever-faster, ever-sharper AFMs has been our group's expertise for many years, and we have already built AFMs that can image beyond 60 frames per second at 256 x 256 resolution - far ahead of commerically available AFMs.
AFM
Our work in imaging bone ultrastructure with the AFM was first published in Hassenkam et al. Bone 35 (2004) 4-10. Please click here to see the beautiful cover of the journal Bone which accompanied our article, and which shows high-resolution AFM imaging of bone.

Figure 1 1 x 1 µm AFM topography images of a fracture surface of a trabecula. The area was rinsed with water that washed away some of the mineral plates that usually coat the collagen fibrils in bone. The collagen fibrils are therefore visible with their characteristic 67-nm spaced banding pattern. The arrows point out aggregates bridging the fibrils in various places, which are probably composed of noncollagenous bone matrix proteins that have, among other complex functions, a mechanical, adhesive role.
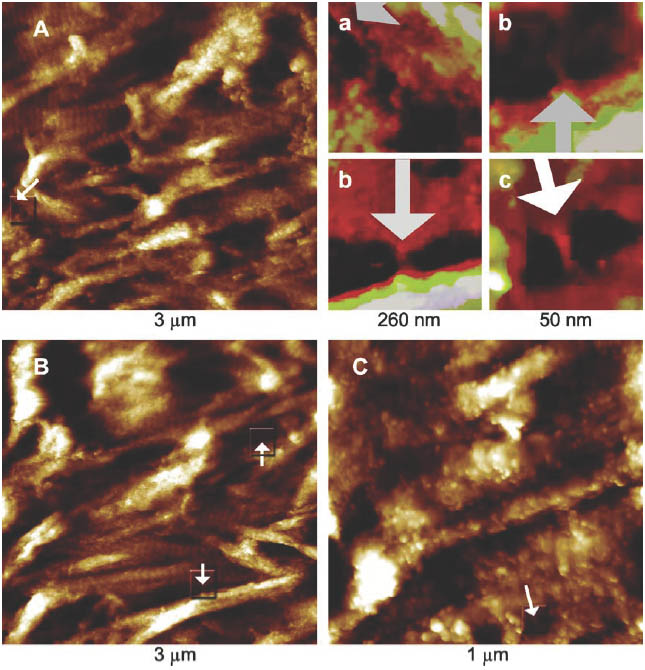
Figure 2 AFM topography images from different regions of the fractured surface of a trabecula that has been partly demineralized by rinsing in water. Here the 67-nm banding pattern in the collagen is evident in all the fibrils. The small protrusions covering the collagen fibrils are probably noncollagenous proteins attached to the fibrils though there may be some remaining mineral plates as well. In some cases, bridging aggregates between individual collagen fibrils is clearly visible, see arrows in the full frame images and in the magnified and computer-enhanced small images (a is from A, b is from B, and c is from C).

Figure 3 AFM image of a fractured bone surface showing bone glue. The fundamental building blocks of bone are collagen-1 fibrils, which are mineralized by being coated with knobbly mineral plates of hydroxyapatite, and a matrix of all many proteins apart from collagen-1 external to the mineralized collagen fibrils, that has many regulatory and mechanical functions. This image shows that the matrix also acts as an interface between mineralized collagen fibrils; the arrows superimposed over this image point to the protein matrix bridging collagen fibrils. Elements of the non-collagen-1 protein matrix have an adhesive role, contributing to the structural integrity of bone. We are studying precisely what those elements are, how they work, and how they fail in aging and disease. (This image was published in our recent paper, Fantner et al. Nature Materials, Vol. 4, p.612-616 (August 2005)).
SEM

Figure 4 SEM image of a microcrack. Only three types of bridges are left spanning this gap - a mostly bare collagen fibril (in the center - its banding pattern can be seen), a mineralized fibril bundle that consists of the same material as the bulk of the bone (beginning from the top left-hand corner) and stringy, unstructured glue-like material (at the left base of the bare collagen fibril, and prominently near the top right-hand corner of this image). It is this glue that is the current focus of our research on bone.
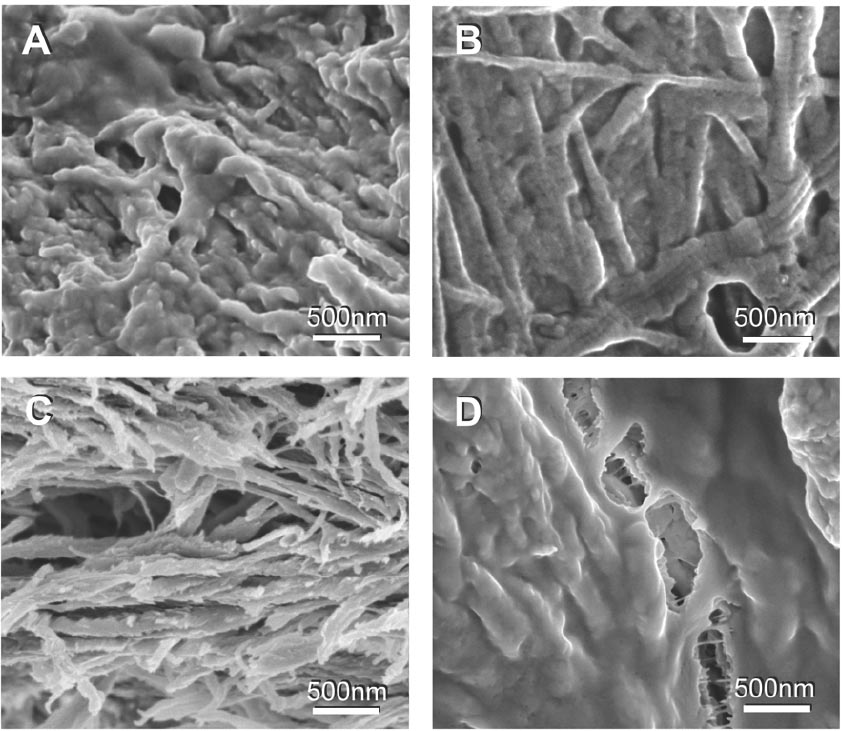
Figure 5 Different morphologies of bone building blocks. Bone is comprised of three basic building blocks - collagen fibrils, mineral plates, and a matrix of unmineralized, non-fibrillar organic material, mostly made of proteoglycans and glycoproteins. (The matrix is also known as the noncollagenous bone matrix). Bone is a highly heterogenous material, partially because it has been adapted to resist different, complex and varying stresses in every species in which it occurs. These scanning electron micrographs show that this heterogenity has its origins in some of the smallest dimensions, as the micrographs were all taken from the same bone sample. They particularly show that the amount of the unmineralized, non-fibrillar organic matrix in trabecular bone varies: A) Fibrils coated with a large amount of non-fibrillar organic material. Particles can still be seen through the smooth cover layer. B) Unmineralized collagen fibrils showing the characteristic 67nm banding pattern. Some particles are between the fibrils but the fibrils are not fully mineralized. C) Mineralized fibrils without non-fibrillar matrix. D) Crack formation in an area with large amounts of non-fibrillar organic matrix. The non-fibrillar organic matrix spans the crack and appears to resist the separation of the mineralized fibrils.
Normal vs aged and diseased human bone
The fundamental question of what makes bone weaker in aging and disease is still not fully understood. We study and compare the effects of aging and disease against the mechanics of normal, healthy bone. Part of our research here is to try to understand how bone quality degrades over time, and particularly whether and how bone glue degrades in aging and disease. Given the recent discovery of bone glue, a study of its properties in aging and disease is a new area of research.
Human bone mass peaks between ages 20-30. A gradual decline in bone mass then ensues for both sexes, until menopause in women, when women begin to drastically lose bone mass. This is one reason osteoporosis is far more common in women and men, but the disease is present in men, too.

Figure 6 SEM of young (22-year old), male human bone

Figure 7 SEM of elderly bone (osteoporotic woman in her '80s)
